- Blog
- Cavalier king charles spaniel rescue chicago
- Terminator t800 quotes
- Michelin map dordogne france
- Winamp windows 10 64 bit free download
- Best free website banner templates
- Gta 6 leak video watch
- Avery label 22806 template for mac download
- 2021 il income tax brackets
- Georgia font free download georgia google font
- Whatsapp desktop video call windows 10
- 3ds max 2015 with keygen
- Bible studies on elijah
- Schedule templates free daily schedule templates free
- Project management plan gantt chart templates
- Wintv v8-5 activation code free
- Michael jackson bad 25 album download zip
- Xbox emulator for pc bluestacks download
- Midnight castle update for android
- Gallant statue animal crossing new leaf
- Abcd of medicare
- Piratebay adobe after effects looks
- Ptsd preschool dsm 5 criteria
- Office word 2013 download free
- Iwrite math 11 solutions
- Arc xt pro download
- Libreoffice breeze dark themes
- Download kodi repositories to usb stick
- Joe bidens first wife daughter death
- Google chrome black out webstore
- Download and installation omnisphere 1
- Butel arc xt pro keys
- Wrestling mpire old school wwf mod
- Professional service invoice template
- Mortgage payment calculation
- Household budget template and tutorial excel
- Business resume template
- Easeus partition master 11-9 license code free
- Water density at 20 c
- Donut blender
- Arduino due vs mega 2560 pinout
- Passport size picture
- The sims 4 cc
- Jdatechooser netbeans 8-2 download
- Free mac mp3 to midi converter
- Blog
- Cavalier king charles spaniel rescue chicago
- Terminator t800 quotes
- Michelin map dordogne france
- Winamp windows 10 64 bit free download
- Best free website banner templates
- Gta 6 leak video watch
- Avery label 22806 template for mac download
- 2021 il income tax brackets
- Georgia font free download georgia google font
- Whatsapp desktop video call windows 10
- 3ds max 2015 with keygen
- Bible studies on elijah
- Schedule templates free daily schedule templates free
- Project management plan gantt chart templates
- Wintv v8-5 activation code free
- Michael jackson bad 25 album download zip
- Xbox emulator for pc bluestacks download
- Midnight castle update for android
- Gallant statue animal crossing new leaf
- Abcd of medicare
- Piratebay adobe after effects looks
- Ptsd preschool dsm 5 criteria
- Office word 2013 download free
- Iwrite math 11 solutions
- Arc xt pro download
- Libreoffice breeze dark themes
- Download kodi repositories to usb stick
- Joe bidens first wife daughter death
- Google chrome black out webstore
- Download and installation omnisphere 1
- Butel arc xt pro keys
- Wrestling mpire old school wwf mod
- Professional service invoice template
- Mortgage payment calculation
- Household budget template and tutorial excel
- Business resume template
- Easeus partition master 11-9 license code free
- Water density at 20 c
- Donut blender
- Arduino due vs mega 2560 pinout
- Passport size picture
- The sims 4 cc
- Jdatechooser netbeans 8-2 download
- Free mac mp3 to midi converter
In general, an increase in temperature may lead to an increase in pressure for a constant volume scenario (e.g., closed container). What is the relationship between temperature and pressure in water? For water, the relationship between temperature and pressure depends on the conditions.
:max_bytes(150000):strip_icc()/smoked-turkey-on-the-grill-336483-Step_05-d28ddbde7d254c928daacf42a26aeb31.jpg)
What is the relationship between density and temperature and pressure? The relationship between density, temperature, and pressure in water is complex and influenced by the thermal expansion and compressibility of the liquid. How to calculate specific volume of water from pressure and temperature? The specific volume of water can be calculated as the reciprocal of density. How do you find the density of water when given pressure? To find the density of water when given pressure, you would need to use the appropriate equation for the compressibility of liquids or refer to tables that provide density values at specific pressure points. How to calculate density at different pressure and temperature? To calculate the density of water at different pressure and temperature values, you can use specialized equations for the compressibility of liquids or consult reference tables. What is the density of water at 20☌ and 1 atm pressure? The density of water at 20☌ and 1 atm (standard atmospheric pressure) is approximately 998.21 kg/m³.
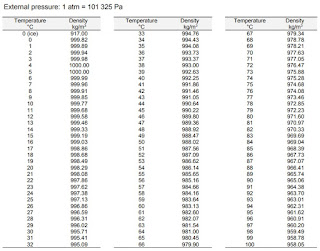
However, the effect of temperature on water density is more significant than the effect of pressure.
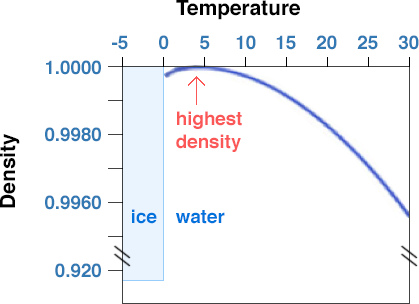
How does pressure and temperature affect density of water? In general, an increase in pressure leads to a slight increase in water density, while an increase in temperature leads to a decrease in water density. However, for most practical purposes, the density of liquid water is relatively constant and can be found in reference tables or using specialized software. How to find density of water at a given temperature and pressure? The density of water at a given temperature and pressure can be calculated using the Ideal Gas Law or the Compressibility Equation for Liquids.
